
DANGERS OF ACTINIC KERATOSIS (AK)
A potential unseen threat of skin cancer1-3,*

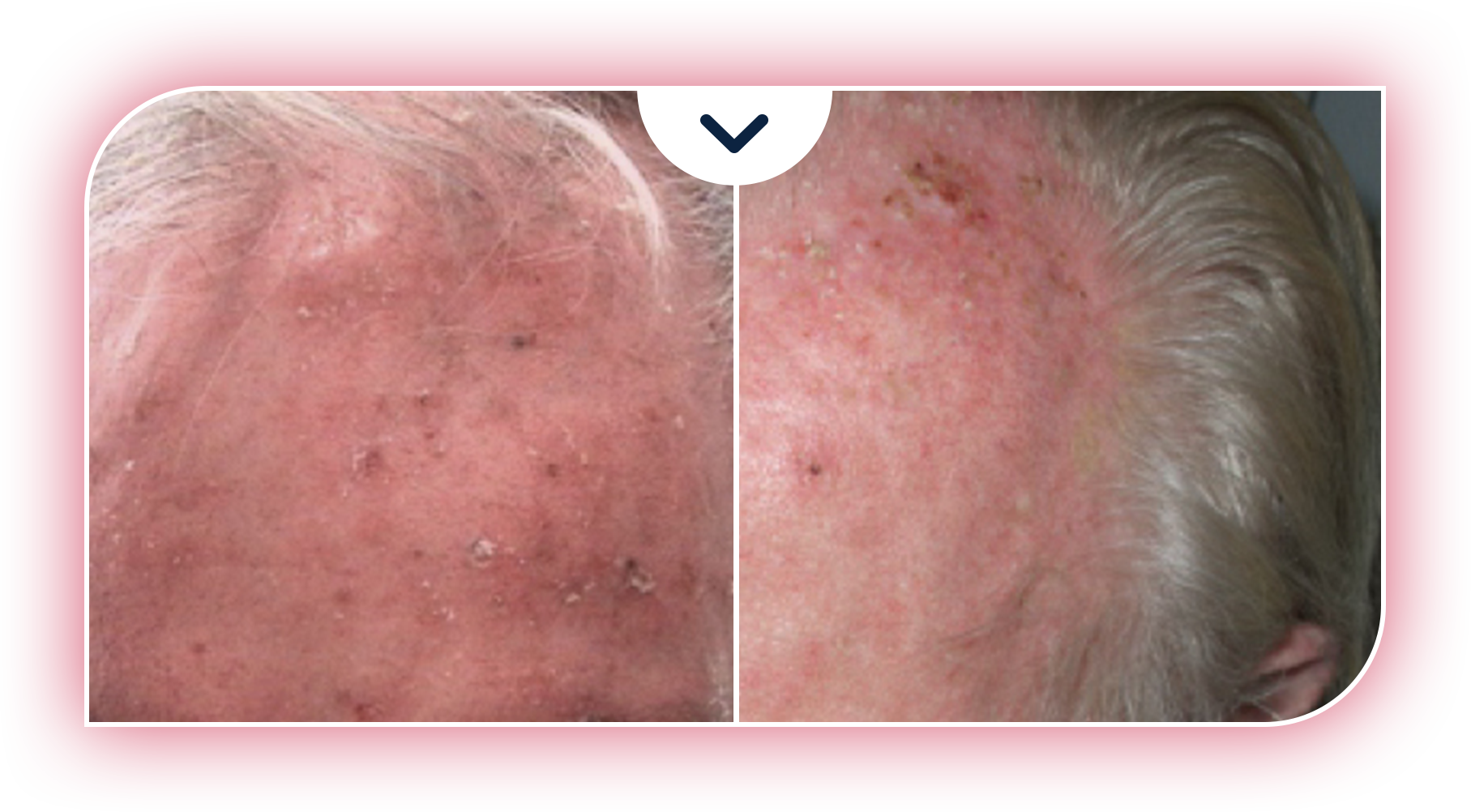
What is actinic keratosis (AK)?
Actinic keratoses are premalignant lesions of the skin that, when left untreated, can potentially develop into squamous cell carcinoma (SCC), the second most common form of skin cancer. AK is primarily caused by chronic sun damage and generally affect people aged 40 or older.1
You could be at risk
- More than a third of the general population over the age of 51 has at least 1 AK lesion that may be found on their head and scalp.4 Those with 10 lesions or more are 3 times more likely to develop SCC. If left untreated, SCC can grow into a more aggressive form called invasive SCC (iSCC).5
What are the signs of AK?
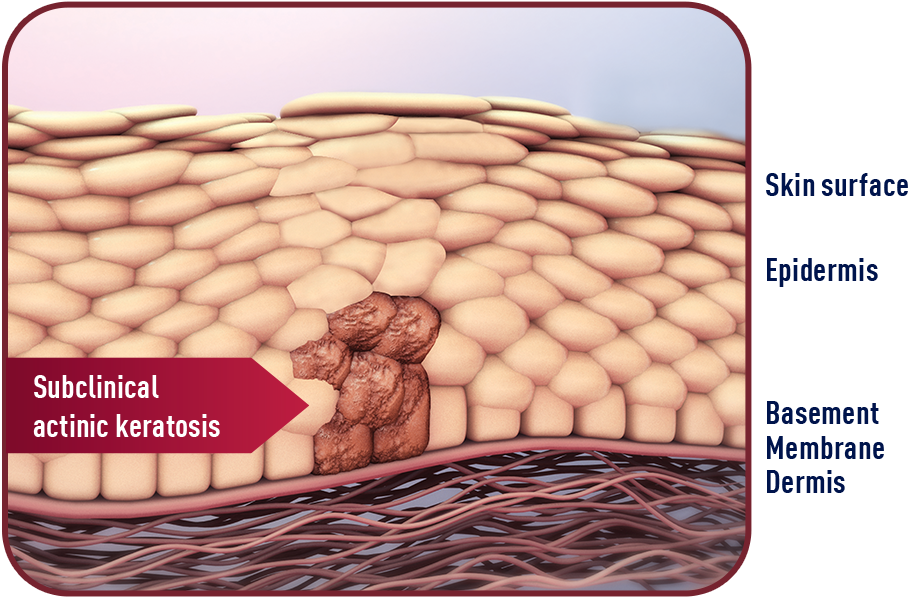
AK lesions typically form on areas of the body most exposed to the sun—such as the face, scalp, neck, and ears. Different types of AK lesions can have a different clinical appearance. AK lesions on the surface of the skin are frequently scaly, range from normal skin color to reddish brown, and can be identified by sight and/or touch. Because AK originates in the skin layers below the visible surface, lesions can be present but still hidden from view. These lesions are known as subclinical lesions.1,3
Sun damage is usually spread over a large area where multiple AK lesions can develop over time. For every visible lesion on the surface, there are likely others hidden below. When surface and subclinical lesions cover a particular area, they form a premalignant field.2,3
If you have concerns you may have AK, please talk with your healthcare provider.

For every AK lesion you can see, there are more you can’t.3



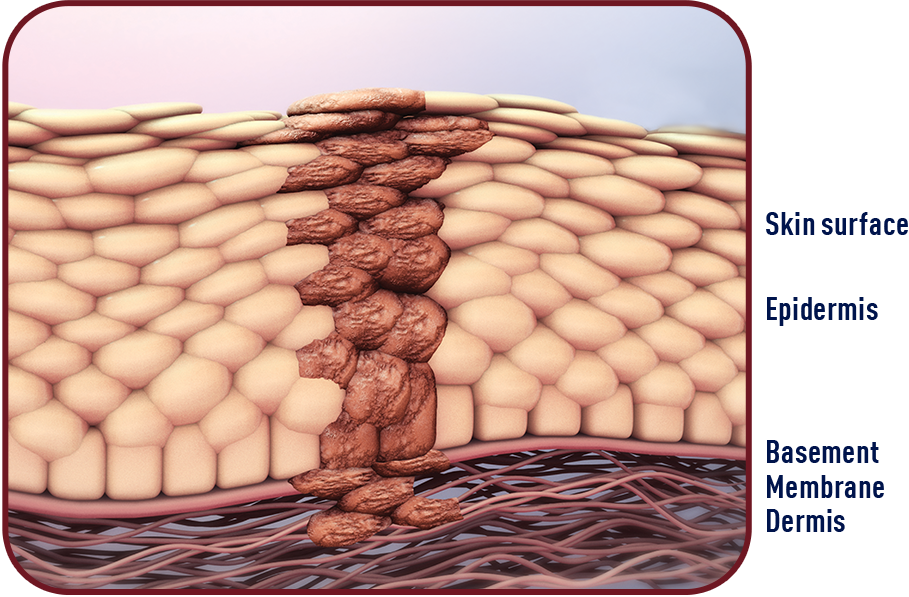
AK may change before you see it.
It’s not uncommon for subclinical AK lesions to progress to iSCC.6 Potential progression risk reported as being between 0.025% and 16%.7
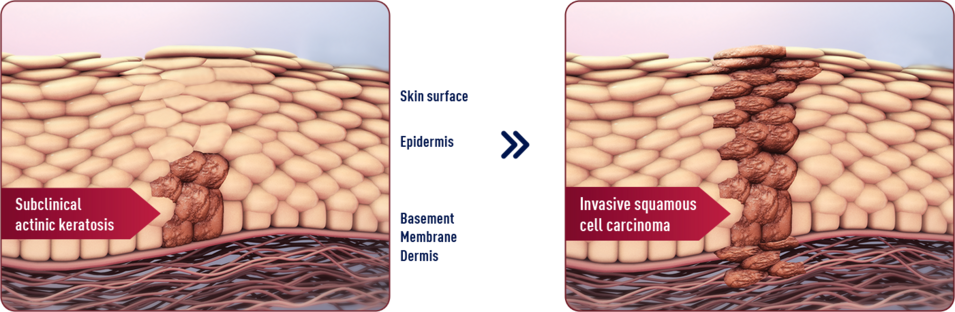
Learn how your healthcare provider can treat mild-to-moderate AK on your face and scalp and address the threat of subclinical lesions.
and BF-RhodoLED®
If you have any questions about AMELUZ® (aminolevulinic acid HCI) topical gel, 10%, we would like to hear from you!
Call us at 1‑844‑4AMELUZ (1‑844‑426‑3589)
Or email us at info‑us@biofrontera.com
DANGERS OF ACTINIC KERATOSIS (AK)
A potential unseen threat of skin cancer1-3,*
What is actinic keratosis (AK)?
Actinic keratoses are premalignant lesions of the skin that, when left untreated, can potentially develop into squamous cell carcinoma (SCC), the second most common form of skin cancer. AK is primarily caused by chronic sun damage and generally affect people aged 40 or older.1
You could be at risk
- More than a third of the general population over the age of 51 has at least 1 AK lesion that may be found on their head and scalp.4 Those with 10 lesions or more are 3 times more likely to develop SCC. If left untreated, SCC can grow into a more aggressive form called invasive SCC (iSCC).5


What are the signs of AK?
AK lesions typically form on areas of the body most exposed to the sun—such as the face, scalp, neck, and ears. Different types of AK lesions can have a different clinical appearance. AK lesions on the surface of the skin are frequently scaly, range from normal skin color to reddish brown, and can be identified by sight and/or touch. Because AK originates in the skin layers below the visible surface, lesions can be present but still hidden from view. These lesions are known as subclinical lesions.1,3
Sun damage is usually spread over a large area where multiple AK lesions can develop over time. For every visible lesion on the surface, there are likely others hidden below. When surface and subclinical lesions cover a particular area, they form a premalignant field.2,3
If you have concerns you may have AK, please talk with your healthcare provider.

For every AK lesion you can see, there are more you can’t.3



AK may change before you see it.
It’s not uncommon for subclinical AK lesions to progress to iSCC.6 Potential progression risk reported as being between 0.025% and 16%.7

Learn how your healthcare provider can treat mild-to-moderate AK on your face and scalp and address the threat of subclinical lesions.
and BF-RhodoLED®
If you have any questions about AMELUZ® (aminolevulinic acid HCI) topical gel, 10%, we would like to hear from you!
Call us at 1‑844‑4AMELUZ (1‑844‑426‑3589)
Or email us at info‑us@biofrontera.com
DANGERS OF ACTINIC KERATOSIS (AK)
A potential unseen threat of skin cancer1-3,*
What is actinic keratosis (AK)?
Actinic keratoses are premalignant lesions of the skin that, when left untreated, can potentially develop into squamous cell carcinoma (SCC), the second most common form of skin cancer. AK is primarily caused by chronic sun damage and generally affect people aged 40 or older.1
You could be at risk
- More than a third of the general population over the age of 51 has at least 1 AK lesion that may be found on their head and scalp.4 Those with 10 lesions or more are 3 times more likely to develop SCC. If left untreated, SCC can grow into a more aggressive form called invasive SCC (iSCC).5


What are the signs of AK?
AK lesions typically form on areas of the body most exposed to the sun—such as the face, scalp, neck, and ears. Different types of AK lesions can have a different clinical appearance. AK lesions on the surface of the skin are frequently scaly, range from normal skin color to reddish brown, and can be identified by sight and/or touch. Because AK originates in the skin layers below the visible surface, lesions can be present but still hidden from view. These lesions are known as subclinical lesions.1,3
Sun damage is usually spread over a large area where multiple AK lesions can develop over time. For every visible lesion on the surface, there are likely others hidden below. When surface and subclinical lesions cover a particular area, they form a premalignant field.2,3
If you have concerns you may have AK, please talk with your healthcare provider.

For every AK lesion you can see, there are more you can’t.3



AK may change before you see it.
It’s not uncommon for subclinical AK lesions to progress to iSCC.6 Potential progression risk reported as being between 0.025% and 16%.7
Learn how your healthcare provider can treat mild-to-moderate AK on your face and scalp and address the threat of subclinical lesions.
and BF-RhodoLED®
If you have any questions about AMELUZ® (aminolevulinic acid HCI) topical gel, 10%, we would like to hear from you!
Call us at 1‑844‑4AMELUZ (1‑844‑426‑3589)
Or email us at info‑us@biofrontera.com
INDICATION AND IMPORTANT SAFETY INFORMATION
INDICATION
AMELUZ® (aminolevulinic acid hydrochloride) topical gel, 10%, a porphyrin precursor, in combination with photodynamic therapy using BF-RhodoLED® lamp, is indicated for the lesion-directed and field-directed treatment of actinic keratoses of mild-to-moderate severity on the face and scalp.
IMPORTANT SAFETY INFORMATION
AMELUZ® (aminolevulinic acid hydrochloride), topical gel, 10%
Purpose: Photosensitizing agent
Uses: AMELUZ® gel, a porphyrin precursor, in combination with photodynamic therapy using BF-RhodoLED® lamp, is used for lesion-directed and field-directed treatment of actinic keratoses of mild-to-moderate severity on the face and scalp.
Warnings:
Do not use if you have a:
- Known hypersensitivity to photoactive substances known as porphyrins.
- Known hypersensitivity to soybeans.
- Known hypersensitivity to any component of AMELUZ®.
Ask your Health Care Provider before use If you have:
- Porphyria (hereditary disease that is characterized by abnormal production of a red blood pigment called heme).
- Photodermatoses (skin conditions caused by or made worse by exposure to light or ultraviolet radiation).
When using this product:
- Allergic reactions: AMELUZ® may cause allergic reactions before photodynamic therapy. AMELUZ® should be washed off and suitable treatment started. The allergic reactions can potentially include severe courses like sudden, severe allergic reaction with breathing difficulty, swelling, lightheadedness, fast heartbeat. sweating and loss of consciousness.
- Transient Amnestic Episodes: Photodynamic therapy may cause transient amnestic episodes (temporary loss of memory). If observed, the therapy must be stopped immediately. If observed after treatment, contact your health care provider.
- Risk of Eye Injury: Patients and health care providers must wear protective eyewear while operating BF-RhodoLED® lamp.
- Photosensitivity: Avoid sun exposure on the treated lesion sites and surrounding skin for approximately 48 hours following treatment.
- Risk of Bleeding: Special care should be taken to avoid bleeding during lesion preparation in patients with inherited or acquired coagulation disorders. Bleeding must be stopped before application of the gel.
- Ophthalmic Adverse Reactions: Avoid applying AMELUZ® into the eyes. Wash eyes with water in case of accidental contact.
- Mucous Membrane Irritation: Avoid direct contact of AMELUZ® with the mucous membranes. Wash with water in case of accidental contact.
- Concomitant use of the following medications may increase the intensity of adverse reactions after light exposure related to photodynamic therapy: St. John's wort griseofulvin, thiazide diuretics, sulfonylureas, phenothiazines, sulphonamides, quinolones, and tetracyclines.
Most common side effects at the application site were:
- skin reddening
- pain/burning
- irritation
- swelling
- itching
- scaling of the skin
- scabbing
- hardening
- blistering
Most side effects occurred during illumination or shortly afterwards, were generally of mild or moderate intensity, and lasted for 1 to 4 days in most cases; in some cases they persisted for 1 to 2 weeks or even longer.
Pregnancy Warning: There is no available data on AMELUZ® use in pregnant women to inform a drug associated risk.
Lactation Warning: There is no available data regarding the presence of the active ingredient (aminolevulinic acid hydrochloride) in human milk or the effects of aminolevulinic acid hydrochloride on the breastfed infant or on milk production.
Pediatric Warning: Safety and effectiveness in pediatric patients below the age of 18 has not been established.
Geriatric Warning: No overall differences in safety or effectiveness were observed between older (65 years and older) and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
Directions:
- AMELUZ® is administered only by a health care provider.
- AMELUZ® is for topical use only.
- Photodynamic therapy with AMELUZ® involves preparation of lesions, application of the product, occlusion and illumination with BF-RhodoLED®.
- Retreat lesions that have not completely resolved 3 months after the initial treatment.
Inactive Ingredients: xanthan gum, soybean phosphatidylcholine, polysorbate 80, medium-chain triglycerides, isopropyl alcohol, dibasic sodium phosphate, monobasic sodium phosphate, propylene glycol, sodium benzoate and purified water.
Other Information:
- Store in a refrigerator, 2°C – 8°C (36°F – 46°F). Excursions permitted to 15°C – 30°C (59°F – 86°F).
- The risk information provided here is not comprehensive. To learn more, talk about AMELUZ® with your health care provider. The FDA approved product labeling can be found at https://Ameluz.com/PI.
- You are encouraged to report side effects of AMELUZ®. Please contact Biofrontera Inc. at 1-844-829-7434 or FDA at 1-800-332-1088 or www.fda.gov/medwatch.
References: 1. Reinhold U. A review of BF-200 ALA for the photodynamic treatment of mild-to-moderate actinic keratosis. Future Oncol. 2017;13(27):2413-2428. 2. Stockfleth E. The importance of treating the field in actinic keratosis. J Eur Acad Dermatol Venereol. 2017;31(Suppl 2):8-11. 3. Berman B, Amini S, Valins W, Block S. Pharmacotherapy of actinic keratosis. Expert Opin Pharmacother. 2009;10(18):3015-3031. 4. Flohil C, van der Leest R, Dowlatshahi E, Hofman A, de Vries E, Nijsten T. Prevalence of actinic keratosis and its risk factors in the general population: the Rotterdam Study. J Invest Dermatol. 2013;133(8):1971-1978. 5. de Berker D, McGregor JM, Mohd Mustapa MF, Exton LS, Hughes BR. British Association of Dermatologists’ guidelines for the care of patients with actinic keratosis 2017. Br J Dermatol. 2017;176(1):20-43. 6. Fernández-Figueras MT, Carrato C, Sáenz X, et al. Actinic keratosis with atypical basal cells (AK I) is the most common lesion associated with invasive squamous cell carcinoma of the skin. J Eur Acad Dermatol Venereol. 2015;29:991-997. 7. Fuchs A, Marmur E. The kinetics of skin cancer: progression of actinic keratosis to squamous cell carcinoma. Dermatol Surg. 2007;33(9):1099-1101.